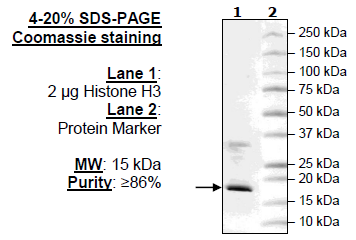
Related targets: SETD2, NSD1, NSD2, H3K36me2, H3K36me3. Hinders histone methylation through suppressing SETD2 and NSD family of methyltransferases.Īpplications: IP, WB, IHC-P, ICC/IF, Flow Cyt, Indirect ELISA.
Histone h3 driver#
H3K36M is a potential oncogenic driver mutation in chondroblastomas also found in pediatric soft tissue sarcomas and HNSCC. IHC of paraffin-embedded human chondroblastoma tissue labeling H3K36M with ab256384 at 1/4000 dilution followed by a goat anti-rabbit IgG H&L (HRP). Related targets: EZH2, SUZ12, EED, p53, PDFRA, FGFR1, ACVR1, BCOR, H3K27me2, H3K27me3 Acts as a suppressor of PCR2 complex and impedes histone methylation.Īpplications: WB, Indirect ELISA, IHC-P, ICC/IF, IP, ChIP H3K27M is found in pediatric brain tumors (pHGG and DIPG), acute myeloid leukemia, melanoma, and glioma. IHC of paraffin-embedded mouse brain tissue labeling H3K27M with ab190631 at 1/500 dilution followed by Discovery UltraMap anti-rabbit (HRP). Furthermore, several studies indicate that replacement of H3G34 with any other amino acid residue blocks SETD2 binding and thereby may hinder methylation of H3K36 1. In GCTB, H3.3G34W expression was shown to promote cellular proliferation in-vitro through inducing transcriptional deregulation and splicing alterations 5. In gliomas, H3.3G34R/V mutations are associated with loss-of-function mutations in the tumor suppressor gene p53 and ATP-dependent helicase ATRX, as well as with the deregulated expression of N-Myc protein 4. While H3G34W mutations are present at high frequency (>90%) in giant cell tumor of bone (GCTB), H3G34R/V mutations are predominantly found in high-grade gliomas of children and adolescents.
Histone mutations at H3G34 include substitutions of glycine 34 with tryptophan (G34W), arginine (G34R), or valine (G34V) ( Figure 1). Genome-wide ChIP-seq analyses suggest that K36M is a driver mutation that alters the epigenetic landscape and promotes neoplastic transformation in the chondrocyte lineage 3.

Similar to H3K27M, the H3K36M mutation suppresses the methyltransferase activity of SETD2 and NSD family proteins and leads to the global reduction of H3K36 di- and trimethylation levels. H3K36M ( Figure 1) is the only recurrent mutation present in the vast majority of chondroblastomas and may act as an oncogenic driver in these tumors. The H3.3K27M mutations are associated with the loss-of-function mutation in p53 and the amplification or gain-of-function mutations in PDGFRA (platelet-derived growth factor receptor), FGFR1 (fibroblast growth factor receptor), and other receptor tyrosine kinases 3. H3K27M mutations have been shown to suppress the methyltransferase activity of the PRC2 complex, leading to a global reduction of H3K27 di- and trimethylation levels (H3K27me2 and H3K27me3) 2. H3K27MĪlthough H3K27M mutations ( Figure 1) are mainly associated with pediatric brain tumors, such as pediatric high-grade glioma (pHGG) and diffuse intrinsic pontine glioma (DIPG), they have also been identified in adult cancers, including acute myeloid leukemia, melanoma, and glioma 1.
Here we review key five histone mutations affecting H3 - H3K27M, H3K36M, H3G34 (W/V/R) - and their role in cancer. The somatic mutations are frequently found in histone 3 (H3), with substitutions of methionine to lysine on H3.3K27 and H3.3K36 being the most common alterations. They are also somatic single-gene mutations that arise at high frequencies in particular diseases. Histone mutations are often heterozygous and frequently comprise single amino acid substitutions in the tails of histone variants. Recurrent histone mutations have been recently described in various cancer types, including brain tumors, chondroblastoma, giant cell tumors of bone, and leukemia. Besides chromatin-associated or regulatory proteins, histone proteins themselves can be mutated in cancers.
